|
In severe cases, people may be affected by high altitude cerebral or pulmonary edema. It typically happens at altitudes above 2,400 meters. This condition can become chronic with prolonged exposure. Some people, such as mountaineers, are affected by altitude sickness, caused by oxygen deficiency in the blood. Travelers, on the other hand, often need to take precautionary measures to avoid discomfort. People and animals, who live at high altitudes, for example in the Himalayas, adapt to the low pressure. The aneroid pressure gauge is based on a pressure sensor - a set of metallic bellows, which change their shape in response to the pressure, which, in turn, rotates the needle by a linkage connected to the bellowsĪtmospheric pressure decreases with the increase in altitude. For this reason, airplane cabins, which would otherwise experience low air pressure at cruising heights, are artificially pressurized. The decrease in atmospheric pressure can cause psychological and physical discomfort for people and animals, or even death. Considerable changes in the atmospheric pressure cause discomfort for people and animals. Atmospheric pressure affects weather and temperature. It usually refers to the weight of the column of atmospheric air above the unit surface area. Atmospheric PressureĪtmospheric or air pressure is the pressure of air in a given environment. Measuring devices often show gauge pressure, although absolute pressure sensors are also in use. This is known as gauge pressure, and it is the pressure measured when determining the air pressure in car tires. In some cases, pressure of gases is measured as the difference between the total or absolute pressure and the atmospheric pressure. The SI unit for pressure is the pascal, which is a newton per square meter. If, on the other hand, you use a blunt knife, you will not be able to cut through because the area is greater and the pressure is lower as a result. The area where the force is applied is small, so the pressure is high enough to cut through the object. For example, if you try pushing a sharp knife down through a carrot or a tomato, you will cut it. 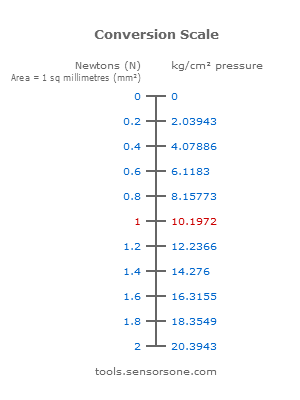
You will probably agree that it is less scary to be stepped on by someone wearing running shoes than by someone wearing stilettos. If the same force is applied to two areas, a smaller and a larger one, the pressure would be greater for the smaller area. Pressure is defined as force per unit of area. Feel free to contact us for any feedback.A balloon bursting at office Lenntech BV is not responsible for programming or calculation errors on this sheet. The constant c is equal to the amount of gas in moles, multiplied by the gas constant: These conditions are often fullfilled, for example for air at atmospheric pressure and standard temperature. This formula is called the ideal gas law.It is valid if the temperature (in kelvin) is at least 50% higher than the temperature at the critical point and the pressure does not exceed the critical pressure. This is because the molecules get more energy and move faster.Ĭ is a constant, proportional to the number of gas molecules If the volume is constant and the temperature increases, the pressure also increases. The pressure of a gas is related to temperature and volume. Relation between temperature, pressure and volume (ideal gas law): If you measure the pressure in Pascal at sea-level, you will find 101325 Pa. At sea-level the pressure is also defined as 1 atmosphere, that's why 1 atmosphere equals 760 torr. It was calibrated that on sea-level the height of the mercury was 760 mm. The reason that mercury is used is that the thermal expansion of mercury is big and is mostly homogeneous. 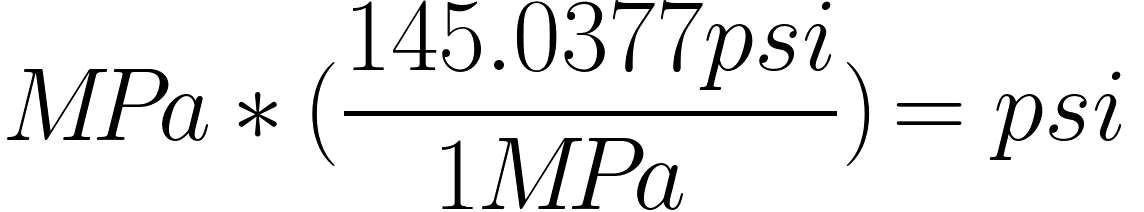 
If the pressure changes, the level of the mercury in the tube changes with it. The unit of pressure used to be torr, also known as mmHG (millimeter mercury). The unit is Pascal (Pa), also known as N/m 2. Pressure ( p) is a derived quantity with its own name. Use this conversion calculator to convert American/Britis units to the metrical (SI) units. Separation and Concentration Purification Request.Plant Inspection & Process Optimalisation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
